Dr. Donath Facial Plastic Surgery in Cincinnati Ohio
COSMETIC
Quantitative Evaluation of Volume Augmentation in the Tear Trough with a Hyaluronic Acid–Based Filler: A Three-Dimensional Analysis
Alexander S. Donath, M.D.
Robert A. Glasgold, M.D.
Jason Meier, M.D.
Mark J. Glasgold, M.D.
Cincinnati, Ohio; Highland Park and
Piscataway, N.J.; and Jacksonville,
Fla
Background: Despite the prevalent use of hyaluronic acid–based filling materials for facial soft-tissue augmentation and favorable reports of durability in the infraorbital region, no quantitative data exist on the long-term durability of these products following injection. This study represents the first attempt to use three-dimensional imaging to quantify augmentation achieved and duration of effect with one hyaluronic acid product in the tear trough.
Methods: The authors conducted a prospective, blinded case series in a clinical setting. One non–animal stabilized hyaluronic acid material was used to augment 20 tear troughs to address cosmetic deficiency in this region. Patients were followed long term with three-dimensional imaging. Posttreatment and pretreatment images were compared, volume change was calculated at each time point, and percentage change between immediate and long-term posttreatment was evaluated. All measurements and calculations were performed independent of the injector.
Results: Residual effect from the hyaluronic acid product was demonstrable on three-dimensional imaging in 100 percent of tear troughs augmented in this study at the final follow-up visit. Average follow-up was 14.4 months (range, 8.5 to 22.75 months). Average initial augmentation measured by three-dimensional imaging was 0.21 cc per site. Average maintenance of effect for patients at the final follow-up visit was 85 percent.
Conclusions: The long-term durability of a small gel particle-based hyaluronic acid in the tear trough is substantiated for the first time in an objective, quantitative fashion using three- dimensional imaging for evaluation of volumetric facial rejuvenation. Three-dimensional photographic imaging offers clinicians a precise and expeditious method for quantitatively evaluating volumetric changes in the face, and represents a significant advance in technology for studying the effects of facial aging. (Plast. Reconstr. Surg. 125: 1515, 2010.)
From Donath Facial Plastic Surgery; Glasgold Group Plastic Surgery; the Department of Surgery, University of Medicine and Dentistry of New Jersey–Robert Wood Johnson Medical School; and Meier Plastic Surgery.
Received for publication May 26, 2009; accepted November 16, 2009.
Copyright ©2010 by the American Society of Plastic Surgeons
DOI: 10.1097/PRS.0b013e3181d70317
Disclosures: The Vectra imaging system used in this study was supplied by Canfield Imaging Systems, Fairfield, New Jersey. No financial support was provided by any corporation, nor was any synthetic filler product provided on a discounted or complimentary basis for this study.
Volume loss in the tear trough imparts an aged, tired appearance to the eye. A growing appreciation for the importance of volume loss has led to a number of techniques to address rejuvenation of this area, including filling with autologous fat or synthetic injectable fillers, fat transposition, or tear trough implants.1 In addition to proper placement and technique, correction of the tear trough requires precise volume augmentation, as even slight overcorrection will result in an unacceptable appearance. In addition, use of fillers in the tear trough is associated with greater morbidity compared with most other facial areas because of an increased incidence and degree of bruising.
Nonetheless, the advent of newer, longer lasting injectable fillers presents an opportunity for excellent results in correction of the tear trough with nonsurgical methods (Fig. 1).
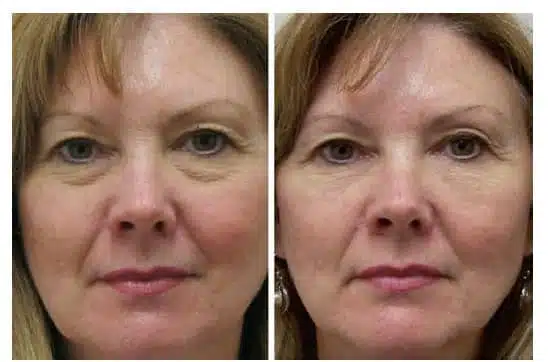
The authors had anecdotally noted in clinical practice that patients treated with small gel particle- based injectable hyaluronic acid filler in the tear trough were demonstrating continued correction at their 6- to 9-month follow-up and beyond. The persistence of volume observed is difficult to document with standard lighting and two-dimensional photographic techniques because a frontal flash will fill the tear trough, obscuring the shadow that would be created with natural or overhead lighting and that cues the observer to the presence of a depression. Three-dimensional imaging reduces the role of lighting in evaluating results in the tear trough region. We report here our experience using the Canfield Vectra-CR 3D camera system (Canfield Imaging Systems, Fairfield, N.J.) to quantitatively evaluate the volume change and duration of result in patients who underwent hyaluronic acid injection augmentation in the tear trough.
The specific injectable hyaluronic acid used in this study was Restylane (Medicis Aesthetics, Scottsdale, Ariz.). The manufacturer of Restylane reports a general duration of effect of approximately 6 months,2 although few studies exist that address the durability of Restylane or other hyaluronic acids specifically in the tear trough region. One such study was presented at the 2005 Annual Meeting of the American Society for Aesthetic Plastic Surgery.3 The majority of patients showed maintenance of full correction at 6 months, and 60 to 70 percent maintained some correction at 1 year. An additional study in 2005 reported durability of 6 to 9 months but did not provide details of analytic methods.4 More recently, Lambros has described persistence of effect to be often in excess of 1 year.5
The majority of reports on the durability of hyaluronic acids in various facial locations, as with those listed above, are based on physician observation, whether directly or by means of examination of two-dimensional photographs.4–11 Our aim was to use the relatively new technology of threedimensional imaging to more objectively quantify the results of soft-tissue augmentation with hyaluronic acids in the tear trough.
PATIENTS AND METHODS
Consecutive patients presenting to a cosmetic surgery practice for rejuvenation of the periorbital region were evaluated for their appropriateness for synthetic filler injection by one of the authors (R.A.G.). Those patients with a history of any treatments in the infraorbital region in the preceding 24 months were excluded from the study, and those with any history of prior volume restoration in this area were excluded. In addition, no patients were admitted to the study who were pregnant or breastfeeding. Twelve patients were identified as candidates for this procedure. Patients were asked to abstain from aspirin, other nonsteroidal antiinflammatory drugs, and other anticoagulant administration for a minimum of 10 days before treatment. After a detailed discussion of risks and benefits, written consent was obtained for off-label.
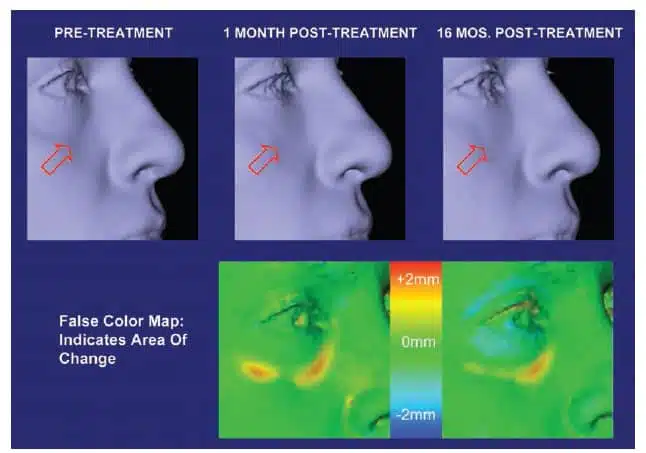
There were no serious adverse events reported. No patients in this study required reduction of volume by means of hyaluronidase injection. Mild treatment-associated adverse events, such as redness, swelling, and ecchymosis, were commonly reported yet transient in nature, typically lasting less than 1 week. No patient exhibited the Tyndall effect (bluish discoloration). All patients reported satisfaction with their results. Subjective diminishment of effect by patients and physician often coincided with objective three-dimensional data. However, at the long-term follow- up visit, patients frequently had limited recollection of their pretreatment appearance and were better able to visualize their volumetric enhancement by means of three-dimensional images versus their own observations or the two-dimensional images. The injecting author (R.A.G.) was consistently able to demonstrate persistence of effect to the patients in this manner until well past 1 year after treatment.
DISCUSSION
This study represents the first long-term, quantitative evaluation of the persistence of hyaluronic acid fillers for facial augmentation.6 Prior studies,4,6 –11 including the recent contribution by Lambros,5 have qualitatively assessed the persistence of various injectable filling materials, and some have included evaluation with the Wrinkle Severity Rating Scale and/or the Global Aesthetic Improvement Scale.9
This study also represents the first of its kind to report the use of three-dimensional photographic imaging in evaluation of volumetric facial soft-tissue filling effects. The ability to quickly and noninvasively create quantitative volumetric measurements of the face is a major advance in technology for studying the effects of facial rejuvenation. The rapid increase in the number of injectable materials being used for facial augmentation, along with disparate and poorly substantiated manufacturer claims regarding the longevity of these materials, create a critical need to produce objective, quantifiable data to allow clinicians to make optimal treatment decisions. This is now possible using a three-dimensional imaging system that has demonstrated an accuracy of 5 to 20 in testing by the National Institute of Standards and Technology.
